WHO Calls for Investigation Into Lab-leak as Possible Coronavirus Origin
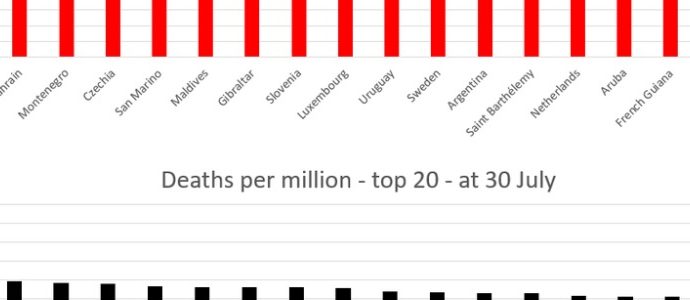
The World Health Organization group tasked with tracking down the origin of the coronavirus responsible for the COVID-19 pandemic is strongly recommending that a deeper investigation be made into the possibility that the SARS-CoV-2 virus may have been released into the public through a laboratory accident. This follows the group’s assertion, afterread more